|
|||||
| |
|||||
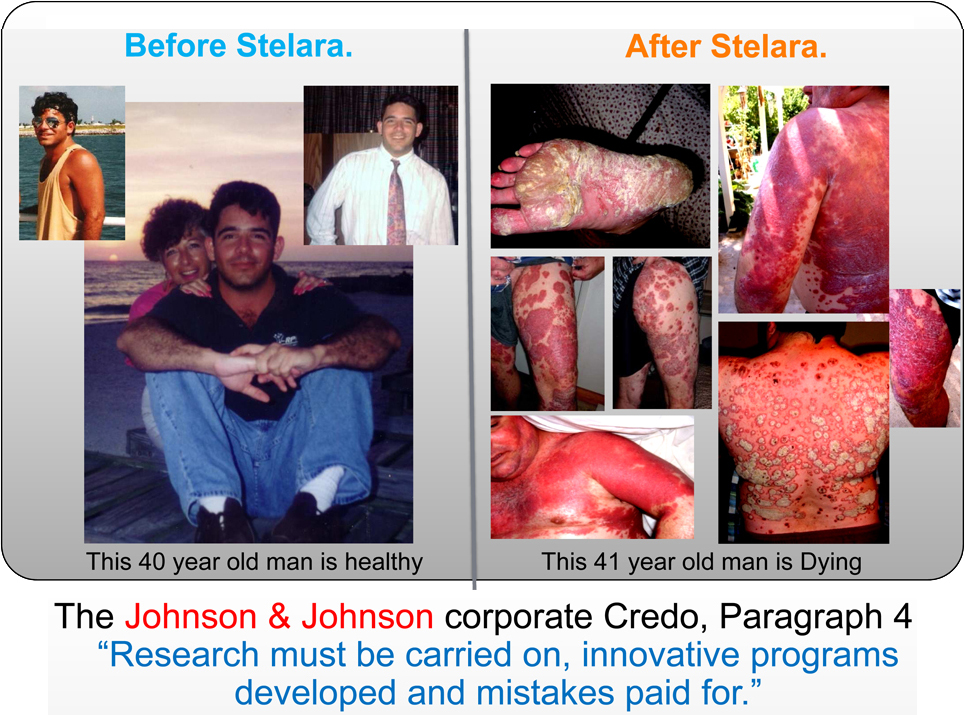
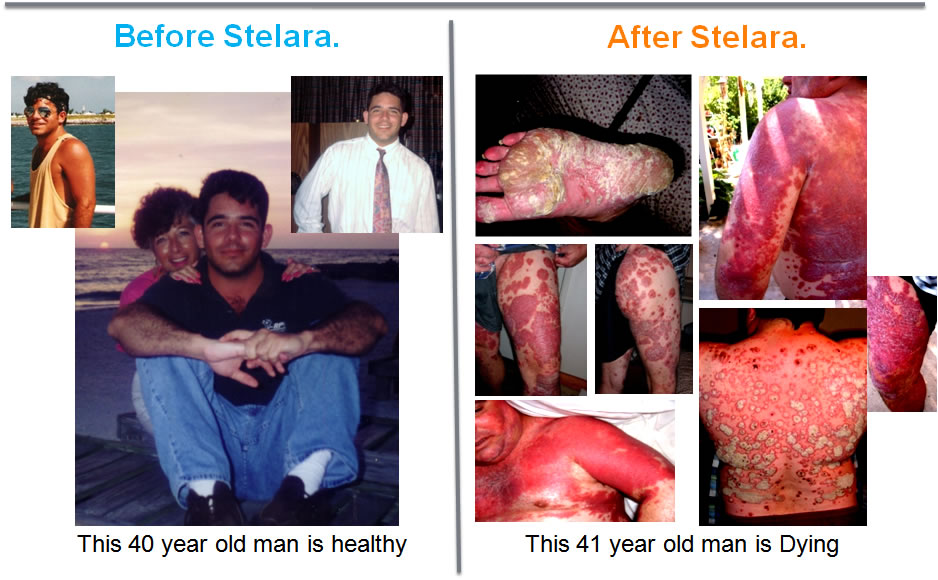
An "FDA APPROVED" drug did this to a healthy man.A man who had only mild Psoriasis for over 7 years.He then took part in a Clinical Trial of Stelara to treat his mild psoriasis. |
|||||
|
|
|||||
|
|||||
|
|||||
(The senate uses web-forms with fields) Use “Health” or “Other” in the subject field”)
Support is appreciated in your own words, or a suggested wording is below, which you can copy and paste. | ||
“Dear Senator, I am a voter in your state. I am very troubled by the situation described at http://www.helpfortony.org/Anthony_Viscosi.pdf. I expect you will take the time to look into this situation. This man is dying because of the negligence and irresponsibility of Johnson & Johnson following a clinical trial for Stelara, in which Mr. Viscosi participated. Neither Johnson & Johnson nor the FDA has offered any help whatsoever to this man as his health deteriorates. Please contact Johnson & Johnson’s CEO, Alex Gorsky at (732) 524-0400 and express your expectation that Johnson & Johnson will make good its written promise to Mr. Viscosi (as presented when he entered its clinical trial program) to fully support him in case of adverse event, and will also follow the Johnson & Johnson corporate Credo, Paragraph 4: “Research must be carried on, innovative programs developed and mistakes paid for.” The Credo was personally written by the leader of the company in 1943; Mr. Gorsky spent a full 30 minutes orating in detail about the Credo at his annual shareholder meeting on April 26th, 2012. Please contact Anthony Viscosi personally at helpfortony@helpfortony.org to express your support. A catastrophic mistake was made with Mr Viscosi. It needs to be remedied. Thank you for your service. Sincerely, Voter" |
||
A shower with Tony. |
||
PARTNERS - Please Contact us at helpfortony@helpfortony.org with any questions or comments |
||