In Britain, the (BHBIA) British Healthcare Business Intelligence Association , British version of the AMA (American Medical Association) and the ABPI, The Association of British Pharmaceutical Industry worked with elected representatives and the industry itself, and simply asked Janssen and all other industry leaders. to sign a common sense set of guidelines for clinical trials. |
||
|
|
||
Live Links: To visit the actual site framed, in full-screen, or download the actual document referenced, click anywhere inside the image. |
||||

|
Full Membership:The top 90% of the industry are signed members of these organizations and their guidelines. |
|||
 |
Janssen:Janssen pharmaceuticals is a signed member of ABPI and The BHBIA. |
|||
|
||||
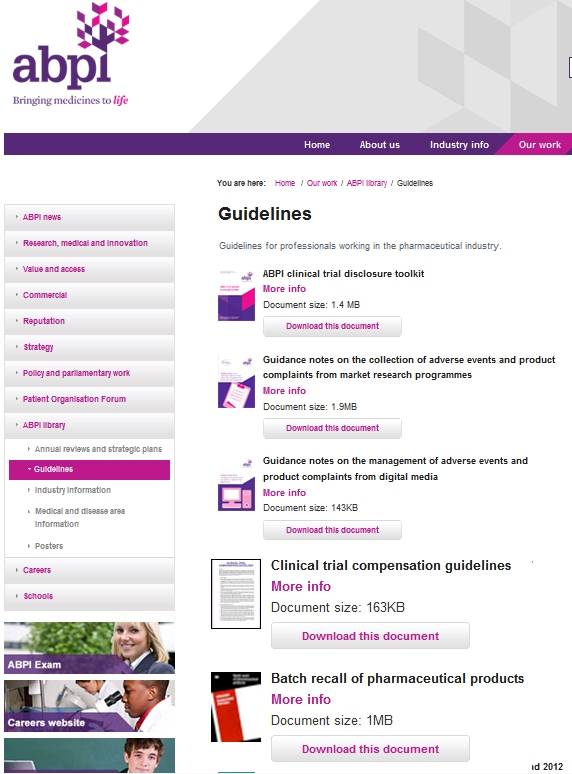 |
The guidelines and agreements.These guidelines were found on the first page of results with the simple search string "Clinical Trials Guidelines". An identiccal search on the AMA web site returned 136 documents. None of them were actual guidelines, rather reports from various meetings held by doctors. |
|||
 |
The Image at left is a sample.The identical real agreement is a simple, straight forward 30 page document that governs all aspects of clinical trials.Click on the image to see the whole genuine document In fact, the portion that details how adverse events are handled is only 3 pages long. (pictured and linked below, left) |
|||
 |
Image:The compensation for adverse events portion (left) is even separated out from the whole agreement and published as its own document on their website.Click on the image to see the entire genuine document. |
|||
So, that is how Britain, Ireland, Scotland and Wales manages clinical trials. We found identical guidelines and procedures for Australia and New Zealand. We will update you as we research other countries. Now The USA, is another matter. |
||||
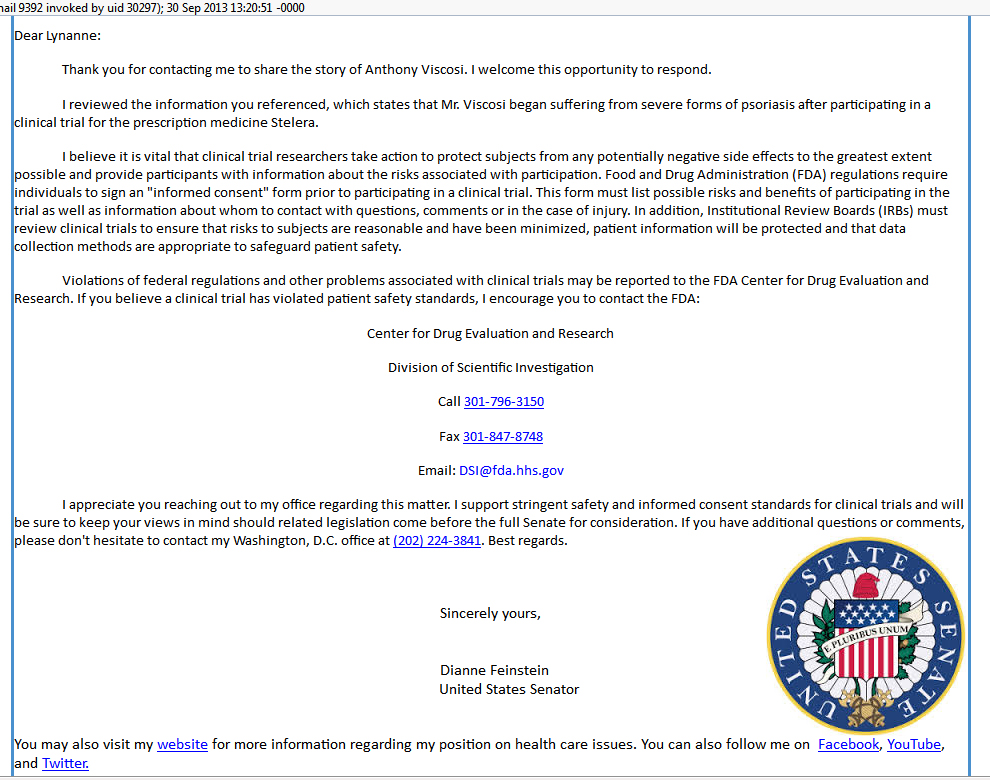
In the U.S.,when a patient in a clinical trial held by Janssen pharmaceutical has an adverse event, Janssen is under no obligation to even provide the patient with information about the drug that ruined their life, let alone provide compensation or medical expertise.
|
||||
Why the huge difference in how the same situation is handled in 2 countries who are considered "sister" countries? |
||||
_________________________In Britain _____________________ |
||||
_________________________In The U.S._____________________
|
||||
In particular, we are focusing on an adverse event that occurred in 2008 during a clinical trial of Janssen's now $1.4 Billion blockbuster drug Stelara.Please visit www.helpfortony.org/action.htm to make a difference. |
||||
 |
||||